Houston Clinical Trial Addresses Growing NAFLD Research Needs
A new clinical trial in Houston is actively recruiting participants with fatty liver disease, representing another significant step in addressing one of the most prevalent liver conditions worldwide. According to ClinicalConnection, this research initiative offers both potential therapeutic benefits for participants and valuable data collection opportunities for the broader clinical research community.
Understanding the NAFLD Research Landscape
Non-alcoholic fatty liver disease (NAFLD) affects approximately 25% of the global population, making it the most common chronic liver condition. The Houston trial comes at a critical time when the clinical research community is intensifying efforts to develop effective treatments for this metabolically-driven disease.
The condition encompasses a spectrum of liver pathology, from simple steatosis to non-alcoholic steatohepatitis (NASH), which can progress to cirrhosis and hepatocellular carcinoma. This progression underscores the urgent need for evidence-based interventions that current research initiatives like the Houston trial aim to address.
Clinical Trial Design Considerations
While specific protocol details for the Houston study remain limited in the initial announcement, fatty liver disease trials typically employ several key design elements that clinical research professionals should consider:
Primary Endpoints
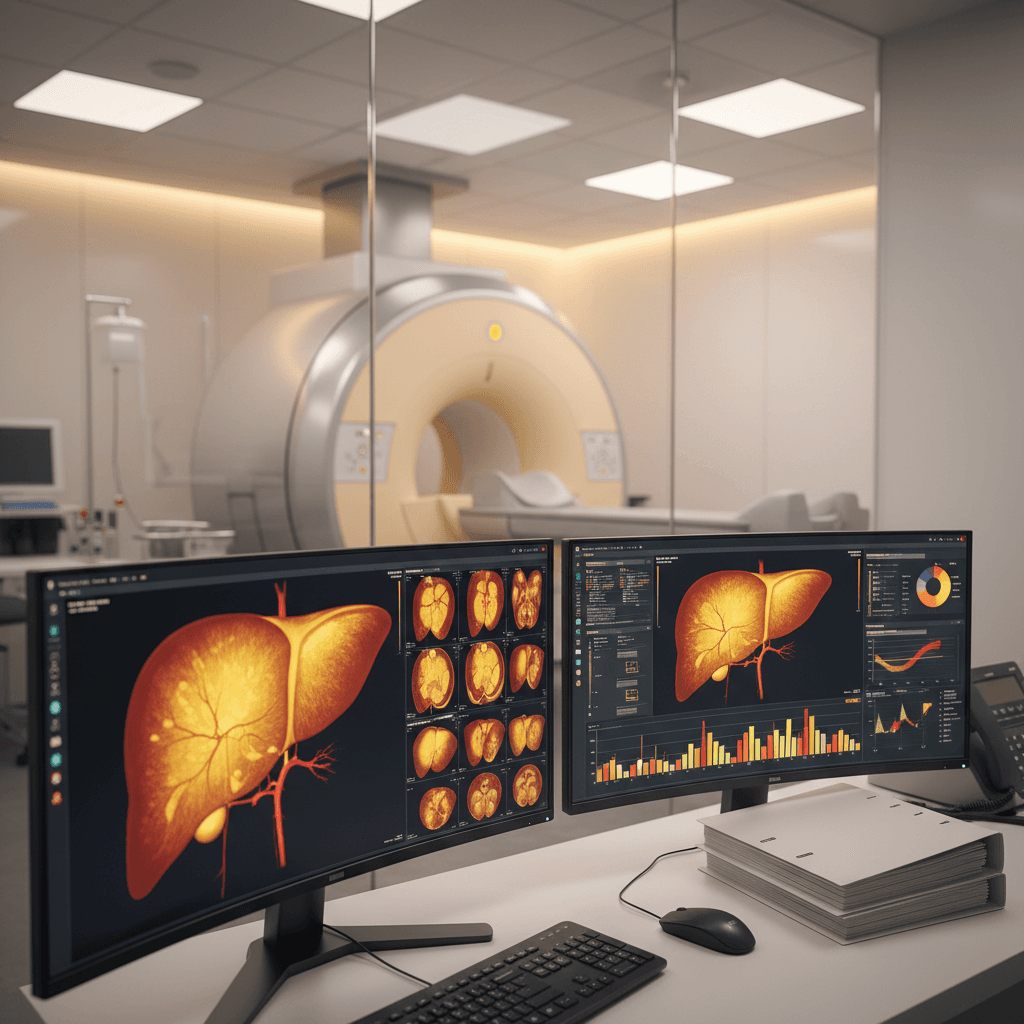
- Hepatic fat reduction measured through MRI-PDFF (Magnetic Resonance Imaging-Proton Density Fat Fraction)
- Histological improvement assessed via liver biopsy when applicable
- Biomarker changes including ALT, AST, and specialized NASH markers
Patient Population Challenges
Recruiting suitable NAFLD participants presents unique considerations:
- Asymptomatic nature of early-stage disease often delays diagnosis
- Comorbidity management requiring coordination with diabetes and cardiovascular care
- Lifestyle intervention compliance affecting trial outcome interpretation
Regulatory and Development Context
The FDA's evolving guidance on NAFLD/NASH drug development has created new opportunities for innovative trial designs. Recent regulatory updates emphasize the importance of clinically meaningful endpoints, which trials like the Houston study help establish through real-world evidence collection.
This regulatory environment parallels other therapeutic areas experiencing accelerated development timelines, similar to the FDA's recent efforts to accelerate mental health drug development, demonstrating the agency's commitment to addressing unmet medical needs across multiple disease states.
Site Selection and Geographic Considerations
Houston's selection as a trial site offers several advantages for NAFLD research:
- Diverse patient population reflecting the ethnic variations in NAFLD prevalence
- Established medical infrastructure supporting complex metabolic research
- Academic-industry collaboration networks facilitating comprehensive data collection
The geographic diversity in clinical trials has become increasingly important, as evidenced by studies like the Atlanta-based gout research, which demonstrates how regional patient populations contribute unique insights to therapeutic development.
Implications for Clinical Research Organizations
For CROs and research sponsors, fatty liver disease trials represent both opportunities and operational challenges:
Operational Considerations
- Specialized imaging requirements for hepatic fat quantification
- Long-term follow-up protocols to assess disease progression
- Integrated care coordination with endocrinology and hepatology specialists
Market Dynamics
The growing NAFLD research market reflects broader trends in metabolic disease drug development. This expansion creates opportunities similar to those seen in other therapeutic areas, such as the competitive landscape emerging in GLP-1 receptor agonist development, where multiple pharmaceutical companies are investing heavily in novel treatment approaches.
Disclosure and Compliance Framework
As with all clinical research, proper disclosure and registration requirements must be met. The FDA's recent reminders to sponsors about ClinicalTrials.gov requirements emphasize the importance of maintaining transparency throughout the research process, particularly relevant for trials addressing widespread conditions like NAFLD.
Future Research Directions
The Houston fatty liver disease trial represents part of a broader research ecosystem working to address metabolic liver disease. Key areas of ongoing investigation include:
- Combination therapy approaches targeting multiple pathogenic pathways
- Biomarker-driven patient stratification for personalized treatment selection
- Digital health integration for lifestyle intervention monitoring
Conclusion
The Houston fatty liver disease clinical trial, as reported by ClinicalConnection, highlights the continued expansion of research opportunities in metabolic liver disease. For clinical research professionals, these studies represent important steps toward addressing a significant unmet medical need while providing valuable insights into trial design, patient recruitment, and regulatory compliance in the NAFLD therapeutic space.
As the research community continues to advance our understanding of fatty liver disease, trials like this Houston initiative will contribute essential data toward developing effective treatments for the millions of patients affected by this condition.
Source: ClinicalConnection - Recent Trials