Critical Product Recall Affects Clinical Research Operations
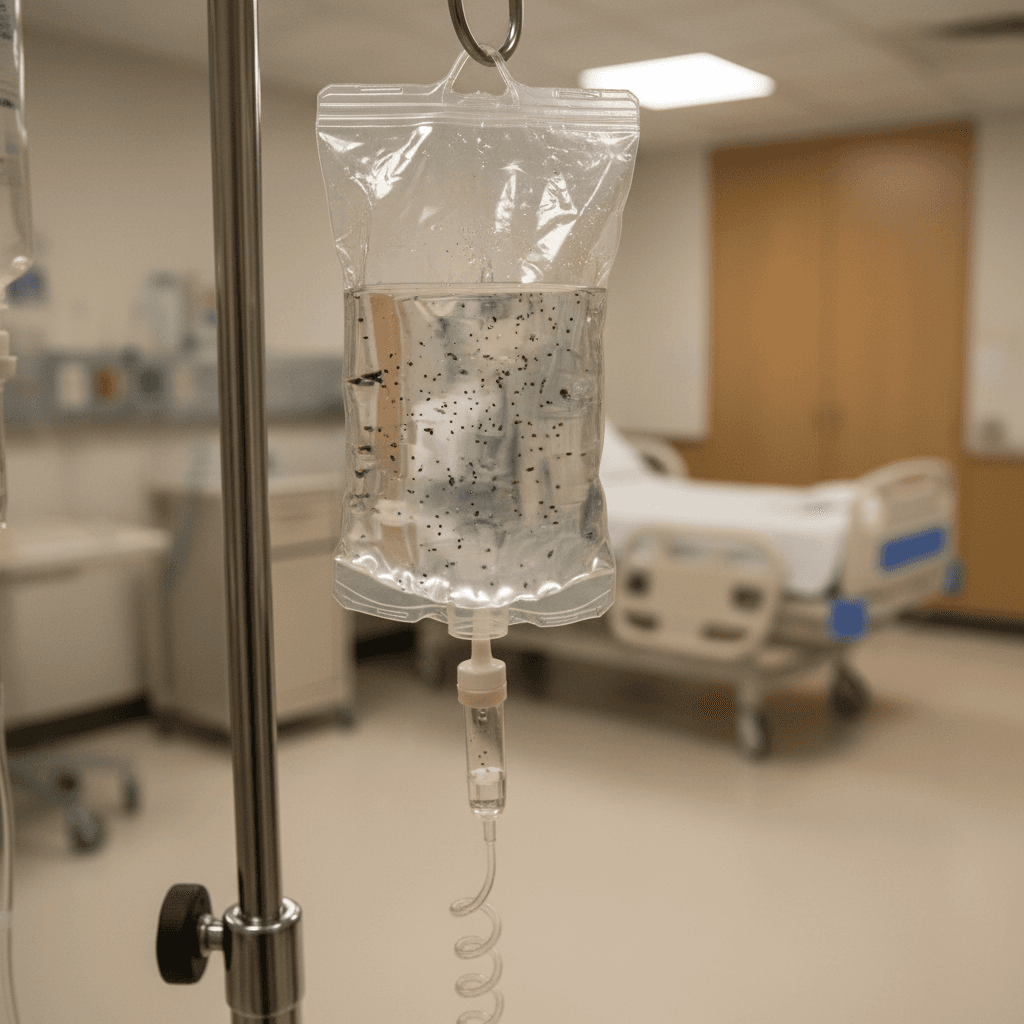
B. Braun Medical Inc. has issued a voluntary nationwide recall of two lots of Lactated Ringer's Injection (E7500, 1L) due to the presence of particulate matter in solution, according to an FDA MedWatch Safety Alert released on April 28, 2026. This recall extends to the hospital and healthcare facility level, potentially impacting ongoing clinical trials and research protocols across the United States.
Understanding the Clinical Significance
Lactated Ringer's injection is a critical isotonic crystalloid solution widely used in clinical settings for fluid resuscitation, electrolyte replacement, and as a vehicle for drug administration. The presence of particulate matter in this essential solution poses significant safety risks, including:
- Embolism risk: Foreign particles can cause vascular occlusion
- Inflammatory responses: Particulate matter may trigger adverse immune reactions
- Infection potential: Contamination could introduce pathogenic organisms
- Equipment malfunction: Particles may obstruct IV lines and infusion devices
Implications for Clinical Research Sites
This recall carries particular significance for clinical research organizations (CROs) and investigational sites that rely on standardized IV solutions for trial protocols. Research teams must immediately:
Protocol Assessment
- Review current study protocols utilizing Lactated Ringer's solution
- Identify affected lot numbers in inventory
- Document any administered doses from recalled lots
- Assess potential impact on study endpoints and safety data
Regulatory Compliance
Clinical research sites must maintain detailed documentation of recall responses, similar to other safety alerts we've covered, such as the TRUE METRIX Glucose Monitor Recall, which demonstrated the importance of swift regulatory response in maintaining trial integrity.
Patient Safety Monitoring
Sites should implement enhanced monitoring for participants who may have received solutions from the affected lots, watching for signs of:
- Fever or inflammatory responses
- Vascular complications
- Unexpected adverse events
- Changes in vital signs or laboratory parameters
Manufacturing Quality Control Concerns
This recall highlights ongoing challenges in pharmaceutical manufacturing quality assurance. Particulate matter contamination typically results from:
- Equipment degradation: Wear of manufacturing machinery
- Environmental contamination: Inadequate cleanroom conditions
- Raw material quality: Compromised source materials
- Process control failures: Insufficient filtration or inspection
The pharmaceutical industry continues to invest heavily in advanced manufacturing technologies and quality systems to prevent such occurrences, but recalls like this underscore the critical importance of robust quality control measures.
Industry Context and Regulatory Response
This B. Braun recall follows recent trends in FDA enforcement of manufacturing standards. The agency has intensified its focus on product quality, as evidenced by recent actions including the Integra LifeSciences Recalls Codman Cranial Drills, demonstrating consistent regulatory vigilance across medical device and pharmaceutical sectors.
The voluntary nature of this recall suggests B. Braun's proactive approach to quality management, likely identifying the issue through internal quality control processes rather than waiting for FDA intervention.
Risk Mitigation Strategies
Clinical research professionals should implement several risk mitigation strategies:
Supply Chain Diversification
- Maintain relationships with multiple IV solution suppliers
- Implement lot rotation protocols to minimize single-lot exposure
- Establish emergency procurement procedures for critical supplies
Enhanced Documentation
- Track lot numbers for all administered IV solutions
- Maintain detailed adverse event logs
- Document all recall-related communications with sponsors and regulatory bodies
Staff Training
- Educate clinical staff on particulate matter identification
- Implement visual inspection protocols before administration
- Ensure proper escalation procedures for quality concerns
Long-term Implications for Clinical Trials
This recall may influence future clinical trial design and execution:
- Protocol amendments: Studies may need to specify acceptable alternative IV solutions
- Supply agreements: Sponsors may require more stringent supplier qualification
- Quality assurance: Enhanced lot testing and inspection protocols may become standard
- Risk management: Improved contingency planning for supply disruptions
Conclusion
The B. Braun Lactated Ringer's recall serves as a critical reminder of the importance of vigilant quality control in clinical research. While voluntary recalls demonstrate industry responsibility, they also highlight the need for robust risk management strategies in clinical operations.
Clinical research professionals must remain vigilant about product quality issues and maintain comprehensive recall response procedures. This incident, like other recent safety alerts in the medical device and pharmaceutical sectors, underscores the dynamic nature of clinical research safety management.
Source: FDA MedWatch Safety Alerts - B. Braun Medical, Inc. Issues Voluntary Nationwide Recall of Lactated Ringer's Injection, April 28, 2026.