
New Schizophrenia Trial Opens in Boston Metro Area: Research Opportunities
Boston-area schizophrenia trial presents new opportunities for clinical research advancement in psychiatric care and patient recruitment strategies.
Daily AI-curated articles on clinical trials, FDA updates, and pharmaceutical insights.

BioNTech announces major restructuring with 1,860 job cuts and site closures in Germany and Singapore, raising questions about future vaccine research capacity.

FDA proposes excluding semaglutide, tirzepatide, and liraglutide from 503B bulk compounding list, citing adequate commercial supply and questioning clinical necessity.
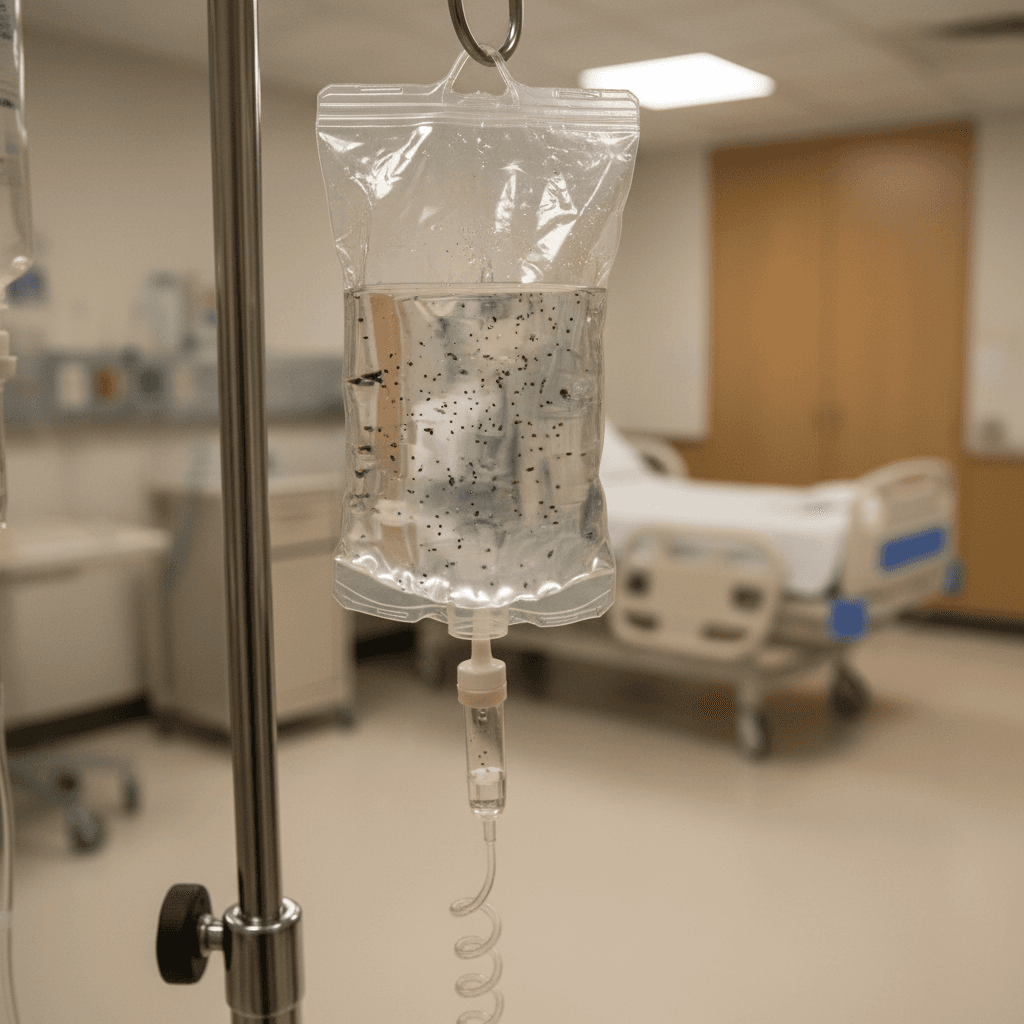
B. Braun Medical recalls two lots of Lactated Ringer's Injection due to particulate contamination, raising critical concerns for clinical research protocols and patient safety.

Eli Lilly's ulcerative colitis study in Kissimmee represents evolving patient-centric recruitment strategies in gastroenterology research, offering insights for CROs.

ArchiMed's acquisition of Esperion Therapeutics signals strategic consolidation in cardiovascular drug development, potentially affecting ongoing clinical trials.

New RAPID pathway promises faster Medicare coverage for breakthrough medical devices, potentially transforming clinical research timelines and patient access strategies.

FDA issues recall alert for Trividia Health's TRUE METRIX glucose monitors due to E-5 error code potentially masking dangerous hyperglycemia in diabetic patients.
New clinical trial in Houston seeks participants with fatty liver disease, offering potential treatments while advancing research in this growing metabolic condition.